Nebula Genomics DNA Report for Myasthenia Gravis
Is myasthenia gravis genetic? We created a DNA report based on a study that attempted to answer this question. Below you can see a SAMPLE DNA report. To get your personalized DNA report, purchase our Whole Genome Sequencing!

Table of contents
What is Myasthenia Gravis?
Myasthenia gravis (MG) is a rare disease that causes weakness in the skeletal muscles needed for movement. It is an autoimmune disorder, which means the body’s immune system attacks healthy tissues and cells instead of foreign invaders like bacteria or viruses. In the case of myasthenia gravis, the immune system attacks the receptors on muscle cells responsible for receiving nerve signals and controlling muscle contractions.
This leads to weakness and fatigue in the muscles involved in the movement, such as those in the eyes, face, throat, and limbs. There are two forms of myasthenia gravis: Ocular and generalized. Normally, the condition called ocular myasthenia gravis starts in the muscles around the eyes causing a noticeable drop in eyelids and difficulty coordinating eye movements.

Once it moves past the eyes it can cause a range of symptoms, including weakness in the arms, legs, and face. This can lead to difficulty chewing, swallowing, speaking, walking, and can cause fatigue that worsens with activity.
The condition becomes especially serious if the respiratory muscles around the chest wall become involved, leading to breathing problems.
In generalized MG, the start often involves not just the eyes, but other parts of the body. A myasthenic crisis occurs if the symptoms become severe enough to cause respiratory failure. About 15 to 20 percent of people with the condition experience at least one myasthenic crisis.
Is Myasthenia Gravis Genetic?
Myasthenia gravis can have a genetic component, but it is not usually inherited. No single gene has been identified as the cause of the condition, but several genetic factors have been associated with an increased risk of developing the condition.
HLA genes: The human leukocyte antigen (HLA) genes are involved in the immune system’s recognition of self and non-self. Certain genetic variants of the HLA genes are more common in people with myasthenia gravis, suggesting that they may contribute to the development of the condition.
PTPN22: The protein tyrosine phosphatase non-receptor type 22 (PTPN22) regulates immune cell function.
CTLA4: The cytotoxic T-lymphocyte antigen 4 (CTLA4) gene regulates immune cell activation.
TNFRSF13B: The tumor necrosis factor receptor superfamily member 13B (TNFRSF13B) gene develops and matures B cells, which produce antibodies.
LRP4: The low-density lipoprotein receptor-related protein 4 (LRP4) gene forms and maintains neuromuscular junctions.
While these genetic factors have been associated with an increased risk of developing myasthenia gravis, not everyone with these gene variants will develop the condition. Other factors, such as environmental triggers, most likely also play a role.
Preganant women who have the disorder may pass antibodies through the placenta, resulting in transient neonatal myasthenia gravis. It generally occurs in only 10%-15% of newborns born to women with the condition.
It should not be confused with congenital myasthenic syndromes, a diverse group of disorders with an underlying defect in the transmission of signals from nerve cells to muscles. These conditions are normally genetic, transmitted by autosomal recessive inheritance.
Epidemiology
Myasthenia gravis is a rare disease, with an estimated prevalence of 14 to 20 cases per 100,000 people worldwide. The condition can affect people of all ages, but it is most commonly diagnosed in women under the age of 40 and in men over the age of 60.
There are also some differences in the incidence and prevalence of Myasthenia gravis in different populations. For example, studies have shown that it is more common in the Americas and least common in Europe.
Myasthenia gravis can also occur in people living with underlying medical conditions or those exposed to certain environmental factors. For example, people with other autoimmune disorders, such as thyroid disease or lupus, may be more likely to develop it. Some studies have also suggested that exposure to certain chemicals or infectious agents may increase the risk of developing Myasthenia gravis.
Symptoms
The symptoms vary depending on the muscles affected. Symptoms may also fluctuate in severity, worsening during stress or after exertion.
Muscle weakness is the most common symptom. It can affect any body part, although it typically starts in the eyes, face, and throat. Weakness can range from mild to severe and be constant or come and go. It may be more noticeable during activity or at the end of the day.
Typically, myasthenia gravis affects the muscles around the eyes first. This can result in both diplopia and ptosis. Double vision (diplopia) causes the individual to see two images of a single object. On the other hand, drooping eyelids (ptosis) can affect one or both eyes and may worsen after prolonged use or as the day progresses.
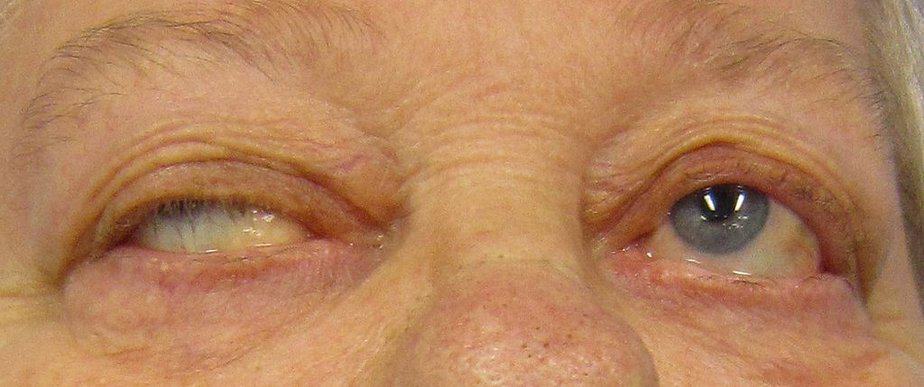
Usually, the condition will spread to other parts of the body, starting with the face and throat. When the muscles used for speaking and swallowing weaken, patients experience difficulty speaking and swallowing. They may have slurred or nasal speech or they may have trouble chewing and swallowing food.
Weakness may then spread to the limbs, which can cause difficulty with walking, standing, lifting, and holding up the head.
If the condition spreads to the muscles needed for breathing, shortness of breath can occur.
Fatigue is also a common symptom of myasthenia gravis and may worsen with activity or as the day progresses.
People with myasthenia gravis are at higher risk for other autoimmune disorders such as autoimmune thyroid disease and systemic lupus erythematosus.
Causes
People who have the condition produce antibodies that block or destroy muscle receptors responsible for communicating through a neurotransmitter called acetylcholine. With fewer receptor sites, muscles receive fewer nerve signals, resulting in weakness.
Some patients also have antibodies that attack a protein called muscle-specific receptor tyrosine kinase, which forms the nerve-muscle junction. Researchers have identified other antibodies as well.
Doctors aren’t able to detect an antibody positive patients in some people who have clinical manifestations of the disease, suggesting there are more antibodies to be discovered.
While the biological cause is known, what brings on myasthenia gravis is not yet fully understood. There do not appear to be risk factors for the condition.
Genetics and environmental factors most likely both contribute. Although myasthenia gravis is not inherited, some people may have genetic variants that predispose them to the condition. Additionally, certain environmental factors, such as viral infection or exposure to certain drugs, can trigger the onset or exacerbate existing symptoms.
The thymus gland, which produces certain immune cells, is often abnormally large in people with myasthenia gravis. This gland is normally large in infancy through puberty and then shrinks until it is much smaller. In many adults with myasthenia gravis, the thymus gland remains large.
Scientists believe the thymus gland in this state also gives incorrect signals to antibodies, causing them to attack the body’s cells.
People with autoimmune diseases, such as lupus or rheumatoid arthritis, are at an increased risk of developing myasthenia gravis.
Diagnosis
Diagnosing myasthenia gravis can be challenging, as the symptoms of the condition can vary widely and can be similar to those of other neurological or neuromuscular disorders. However, several diagnostic methods exist to determine if someone has the condition.
First, a healthcare professional will typically perform a physical exam to assess the individual’s muscle strength and look for any visible signs of weakness, such as drooping eyelids or difficulty swallowing. Neurological tests may include reflexes, muscle strength, muscle tone, senses of touch and sight, coordination, and balance. If a person has the condition, their muscle fibers will not respond as well to stimulation.
Cooling muscles affected by myasthenia gravis may transiently improve weakness. This test is simple and easily performed on a droopy eyelid, often helping to confirm a diagnosis.
Blood tests can be used to check for abnormal antibodies, including acetylcholine receptor antibodies (AChR) and muscle-specific kinase (MuSK) antibodies.
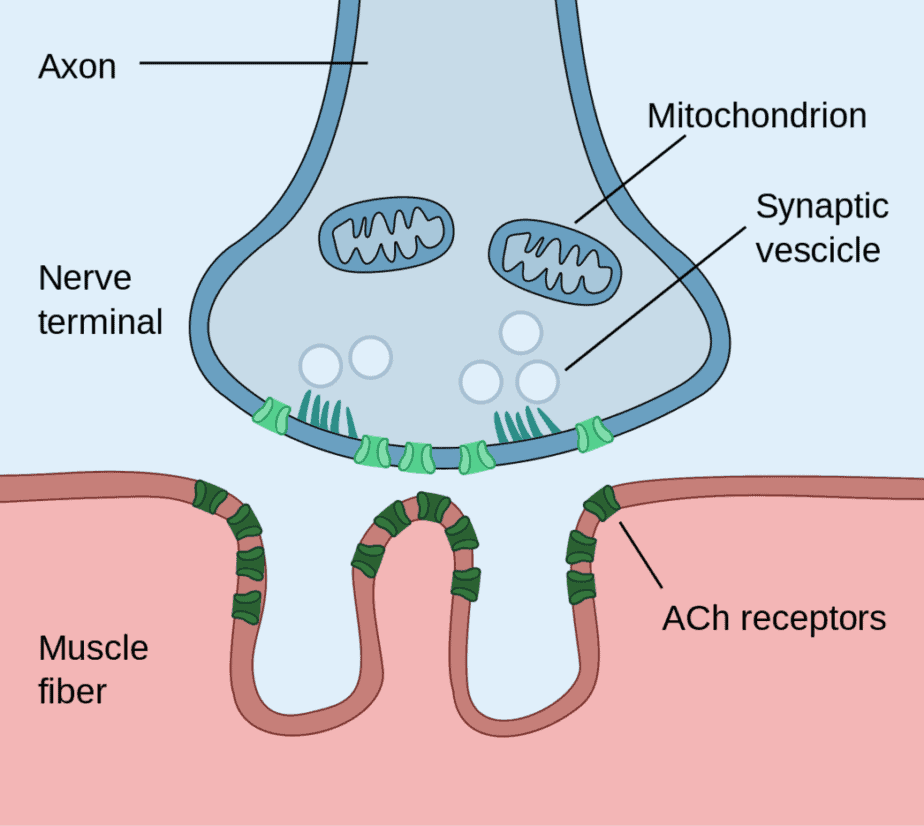
A repetitive nerve stimulation study involves the delivery of small electrical impulses to the nerves that control muscle movement. In patients with myasthenia gravis, the response of the muscles to the electrical impulses may decrease over time. Another nerve test, electromyography (EMG), involves the placement of small electrodes on the skin that can measure the electrical activity of the muscles.
Imaging tests such as CT scans or MRIs may be used to look for a tumor or abnormalities in the thymus gland.
Treatment
While there is no cure for myasthenia gravis, several treatment options can help reduce symptoms and improve the quality of life. The specific treatment for myasthenia gravis will vary depending on the individual’s symptoms, the severity of the disease, and other factors.
Medications
Medications that increase the availability of acetylcholine, a neurotransmitter that plays a crucial role in muscle movement, can help improve muscle strength and reduce fatigue. These medications include cholinesterase inhibitors, such as pyridostigmine, immunosuppressive drugs, and corticosteroids. Side effects of long term use can include stomach problems, weight gain, mood swings and an increased risk of infections.
Intravenous treatments
Plasmapheresis: This procedure is similar to dialysis. It involves the filtration and removal of antibodies in the blood responsible for attacking the muscle receptors. This treatment can provide temporary relief, but usually for only a few weeks.
Intravenous immunoglobulin (IVIg): This treatment involves infusing normal antibodies, designed to block the harmful antibodies that attack the muscle receptors. It is usually affective for three to six weeks.
For those who don’t respond to other treatments, doctors may recommend antibody therapy with Rituximab (Rituxan) or eculizumab (Soliris).
Surgery
If your doctor finds a tumor in the thymus gland (thymoma) they may recommend surgical removal of the thymus. A thymectomy may help improve symptoms and reduce the need for medication. A surgeon may perform either an open or minimally invasive surgery. The latter usually involves small incisions and guided removal of the thymus gland that may include video or robot-assisted technology.
Various supportive therapies may be used to help manage symptoms and improve quality of life. These may include physical therapy to improve muscle strength and mobility, speech therapy to improve swallowing and speech, and respiratory therapy to help with breathing difficulties.
The specific treatment plan will vary depending on the individual’s symptoms and medical history. Additionally, most people can manage their symptoms and lead relatively normal lives with appropriate treatment. However, it is a chronic condition that requires ongoing management and monitoring.
For more information you may want to consult the National Institute of Neurological Disorders and Stroke (NINDS) for the latest information.
If you liked this article, you should check out our other posts in the Nebula Research Library!
June 16, 2023
