DNA polymerase definition
DNA polymerases are enzymes that catalyze the synthesis of DNA molecules from deoxyribonucleotides. DNA polymerases play a key role in DNA replication enabling passing of genetic information to daughter cells from generation to generation.
Edited by Christina Swords, Ph.D.
Biochemical aspects of DNA polymerases
Polymerase activity
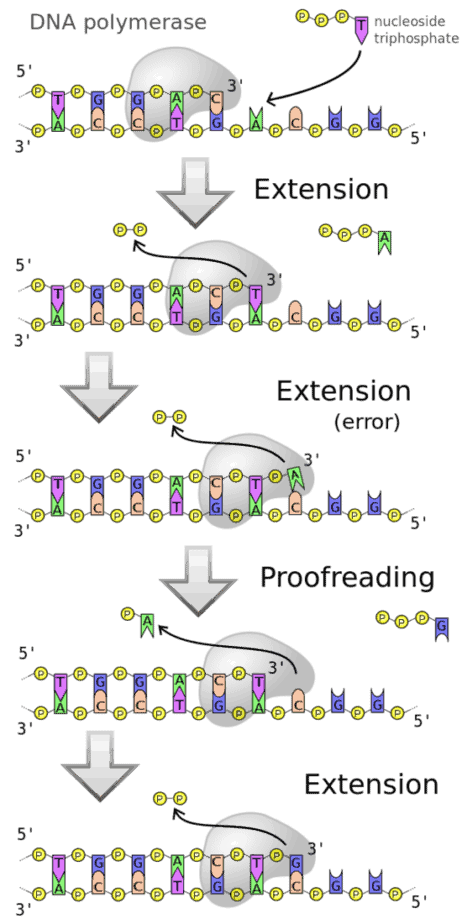
The polymerase enables the chemical linking of individual molecules (monomers) to form a chain (polymer). In the case of the DNA polymerase, the polymer formed is deoxyribonucleic acid (DNA). The monomers are deoxyribonucleotides, more precisely deoxynucleoside triphosphates (dNTPs). The DNA-dependent DNA polymerase always uses an already existing DNA single strand as a template for the synthesis of a new, complementary strand whose nucleotide sequence is thus determined by the template. This preservation of the DNA sequence is decisive for the ability of the DNA polymerase to copy the genetic information encoded in the DNA. The correct copying of the template is achieved by complementary base pairing of the incorporated nucleotide bases with the bases of the DNA template, mediated by hydrogen bonds. The synthesis of the new DNA strand takes place from the 5′ to the 3′ end. Chemically, a nucleophilic attack of the terminal 3′-hydroxy group of the DNA strand on the α phosphate of the dNTP takes place, releasing pyrophosphate. This step is catalysed by the polymerase.
In contrast to RNA polymerases (produces RNA that is used to synthesize proteins from amino acids), the synthesis of the complementary DNA strand in DNA polymerases can only take place if a free 3′-hydroxy end is available to the polymerase. The first nucleotide is then attached to this end. In the polymerase chain reaction (PCR), a DNA single strand (primer) of about 15-20 nucleotides in length is used as the starting point of the reaction. The enzymes usually require magnesium ions as cofactor.
The catalysis of the formation of the diester bond is functionally analogous to the corresponding reaction of RNA polymerases. The last nucleotide of the section already synthesized and the nucleotide to be added are coordinated to one of two magnesium ions each in the catalytic center of the polymerase domain. The first phosphate group of the nucleotide to be added is coordinated to both magnesium ions. The spatial position allows the hydroxy group of the preceding nucleotide to attack the phosphate group of the nucleotide to be added. In the process, a pyrophosphate residue is split off.
Exonuclease activity
Many polymerases also have other enzyme functions. In the presence of low concentrations of dNTPs, the 3’→5′ exonuclease activity for nucleotide removal predominates. Some polymerases also have 5’→3′ exonuclease activity. To ensure that no errors occur when reading the DNA template, they have this proof reading function, i.e. they are able to detect the insertion of an unsuitable nucleotide and then remove it from the DNA by means of the exonuclease activity. This enables the degradation of an existing DNA or RNA strand that is already paired with the template strand while a new strand is being formed. This results in an exchange of the old strand for a new strand. This exonuclease activity is exploited by the nick translation method.
Different DNA polymerases
In bacteria such as Escherichia coli there are three different DNA-dependent DNA polymerases. One of them, DNA polymerase I (Pol I) was isolated in 1955 by Arthur Kornberg and was the first polymerase ever discovered. However, this is not the most important polymerase for replication in E. coli, as it only catalyses about 20 synthesis steps (i.e. it has only a low processing power). However, it is responsible for primer degradation during replication due to its 5’→3′ exonuclease activity. DNA polymerase II and DNA polymerase III, the other two DNA polymerases in E. coli, were isolated only 15 years after the discovery of DNA polymerase I, after E. coli mutants with a defect in the polymerase I gene nevertheless proved to be competent for replication. However, these mutants were particularly susceptible to UV radiation and alkylating substances, which is why it is assumed that DNA polymerase I mainly performs repair tasks. Polymerase III, which carries out the actual replication in E. coli, is composed of a total of seven subunits and only occurs in very few copies per bacterial cell.
Eukaryotic DNA polymerases, including human DNA polymerases, are classified into the following families:
- Family A: DNA polymerases γ, θ and ν
- Family B: DNA polymerases α, δ, ε and ζ
- Family X: DNA polymerases β, λ, σ and μ
- Family Y: DNA polymerases η, ι and κ
The polymerase γ only occurs in mitochondria.
Only five types occur in mammals: α, β, γ, δ and ε. It is assumed that the polymerases δ and ε, which are decisive for replication, are characterized by high processing power and proof reading function. In contrast, the polymerases α and β show only low processing power and no proof reading function.
Furthermore, there are RNA-dependent DNA polymerases that use RNA as a template and attach dNTPs to it. These are called reverse transcriptases, which also includes telomerase. The only known independent DNA polymerase is the terminal deoxyribonucleotidyltransferase.
In archaebacteria there are temperature-stable types that are also used for PCR.
Biological significance
DNA polymerases are of central importance for DNA replication. They enable the faithful copying of genetic information in the form of DNA, thus a decisive step in the reproduction and procreation of living organisms. The enzymes also play an important role in processes associated with DNA repair.
Biotechnological significance
In the laboratory, DNA polymerases are often used for the polymerase chain reaction and related methods (e.g. RT-PCR, qPCR), for nick translation, random priming and DNA sequencing. A large number of different thermostable types (e.g. Taq polymerase from Thermus aquaticus) are used, some of which are modified by protein engineering. In addition to high temperature stability, thermostable DNA polymerases of archaic origin, such as Pfu polymerase, provide proof reading, since the PCR should not lead to any changes in the DNA produced. Furthermore, strand displacing DNA polymerases such as φ29 DNA polymerase are used in various methods of isothermal DNA amplification at room temperature. The precursor of the DNA polymerases used today was the T4 DNA polymerase.
Did you like this blog post? You can find more post about DNA, genetics and sequencing here!
You might be interested in the following related blog posts:
