Nebula Genomics DNA Report for Crohn’s Disease
Is Crohn’s disease genetic? We created a DNA report based on a study that attempted to answer this question. Below you can see a SAMPLE DNA report. To get your personalized DNA report, purchase our Whole Genome Sequencing!

Table of contents
What is Crohn’s Disease?
Crohn’s disease is a chronic inflammatory bowel disease (IBD) that damages the digestive system. Doctor’s characterize the condition as a decreased ability of certain cells to recognize and fight bacteria, an altered mucus layer on the intestinal mucosa, and increased permeability of the intestinal epithelium.
Chronic inflammation in the digestive tract can even lead to normal cells being replaced by scar tissue. Although it can affect any part of the gastrointestinal (GI) tract from the mouth to the anus, inflammation is most frequent in the lower small intestine and also affects the colon and large intestine. Less frequently, it may affect the esophagus and the mouth.
Common symptoms include abdominal pain, severe diarrhea, fatigue, weight loss, and malnutrition. The condition evolves and spreads as it progresses. However, is most commonly expressed as periodic flares rather than a continuous condition.
If left untreated, people with Crohn’s disease often experience painful and debilitating symptoms. Sometimes, the condition can even lead to life-threatening complications.
There’s no cure for Crohn’s disease, but there are treatments to help patients manage the symptoms. This is why it is important to contact your doctor if you suspect you may have the condition.
Some therapies are effective enough to induce long-term remission and healing. If treated, patients are able to live fairly unaffected lives. However, if left untreated, the condition can become serious.
Is Crohn’s Disease Genetic?
Many of the major genes associated with the condition are those that code for proteins associated with immune system function. Specifically, they aid the immune system in identifying and responding to various types of bacteria (harmful, neutral, and beneficial) in the lining of the digestive tract. Many of the proteins play roles in autophagy, which is a process that cells use to surround and destroy bacteria and viruses.
MedLine Plus notes some of the more common variants that contribute to Crohn’s disease:
NOD2: This gene codes for a protein that is active in immune cells (monocytes, macrophages, and dendritic cells, etc.). The protein is also present in several types of epithelial cells that line the intestine and plays a role in autophagy.
ATG16L1: This gene codes for a protein that plays an important role in autophagy. This process involves the body’s inflammatory response in addition to removing harmful invaders.
IL23R: This gene codes for the interleukin 23 (IL-23) receptor. Found on the outer membrane of immune cells. When this receptor binds to its protein IL-3, it starts a signaling pathway through the immune cells that prompts an inflammatory response.
IRGM: This gene codes for a protein that helps trigger autophagy in response to certain bacteria.
In total, research has identified at least 200 genetic risk factors connected to the risk of Crohn’s disease. The genetic interactions are complex, and most experts believe that specific genetic mutations account for only a small percentage of disease prevalence.
Current Research on Crohn’s Disease
A paper published in 2017 reports on how Crohn’s disease is influenced by genetics. This study explains how genetic factors are responsible for the pathological symptoms of the condition.
In order to get a significant and accurate result, the researchers studied more than 600 Crohn’s disease patients. Overall, they identified and analyzed three genetic markers associated with Crohn’s disease: MHC, NOD2, MST1. These genes are all located in different sites that can be influenced by the alternation of genetic components and may be individually or collectively responsible for the different types of Crohn’s disease (ileal, ileocolonic, and colonic).
Ultimately, the outcome of this study showed that individuals with these genetic markers are susceptible to Crohn’s disease.
The association of genetic factors to Crohn’s disease was also confirmed by genome-wide association studies (GWAS). In this study, pieces of multi-dimensional data from GWAS, transcriptome-wide association studies, and methylation studies were analyzed by Mega-analysis of Odd Ratio (MegaOR), a method developed by the research group to prioritize associated genes. Overall, this study identified genes associated with Crohn’s disease with a high level of confidence from multidimensional evidence.
The genetic architecture of Crohn’s disease was also studied in patients with Asian, Japanese backgrounds. In this research, the study authors analyzed genetics of 81 patients with Crohn’s disease with about 200 healthy individuals acting as a control. The study also involved patients with ulcerative colitis, which is also a type of inflammatory bowel disease (IBD).
The primary aim of this study is to investigate the SMAD family genes in relation to Crohn’s disease. The result positively identified that an alteration in genetic variants may cause Crohn’s disease in Japanese patients.
Epidemiology
According to the Centers for Disease Control and Prevention (CDC), an estimated 1.3% of US adults (3 million) reported a diagnosis of IBD in 2015, which includes both Crohn’s disease and ulcerative colitis. This number reflects patients diagnosed at age 18 or older, which reflects the fact that most diagnoses occur when patients are in their 20s and 30s.
The disease occurs most often in western Europe and North America. Doctors diagnose as many as 70,000 new cases of IBD in the United States each year. The condition seems to affect men and women at the same rate.
This condition should not be confused with irritable bowel syndrome (IBS), which is a separate condition of the colon that usually does not cause ulcers or lesions characteristic of IBDs.
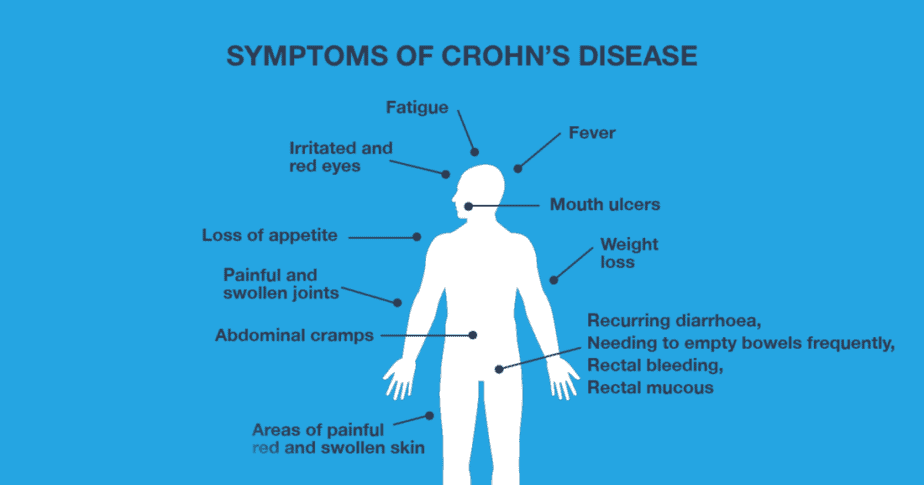
Symptoms
Crohn’s disease affects each patient differently. The part of the GI tract affected often determins the symptoms. The condition is a chronic disease. This means that patients often experience flares of symptoms followed by periods of remission, when no symptoms are present.
The Crohn’s and Colitis Foundation provides a handy resource of symptoms to note if you suspect you have the condition. Some common signs and symptoms include:
- Persistent diarrhea
- Rectal bleeding
- Urgent need for bowel movements
- Abdominal cramps and pain
- Sensation of incomplete bowel evacuation
- Constipation, which can lead to bowel obstruction
- Appetite loss
- Weight loss
- Low energy and fatigue
- Slow growth and development in children
Some serious complications of Crohn’s disease include intestinal blockages, fissures, fistulas, and strictures. A fissure is a tear in the lining of the anus, a fistula is an abnormal channel in the digestive tract caused by inflammation, and a stricture is narrowing of the intestine.
Some of the most common signs that prompt people to contact a healthcare professional include:
- Abdominal pain
- Blood in your stool
- Nausea and vomiting
- Ongoing bouts of diarrhea that don’t respond to over-the-counter (OTC) medications
- Unexplained fever lasting more than a day or two
- Unexplained weight loss
Causes
The exact cause of Crohn’s disease is unknown, but doctors suspect a combination of hereditary, genetic, and environmental factors plays a role. Although diet and stress may aggravate the condition, they do not cause the disease (as was previously thought).
The symptoms of Crohn’s disease are ultimately caused when the immune system mistakes harmless bacteria in the digestive tract for harmful foreign substances. As a result, the bacteria trigger an immune response that creates chronic inflammation. During this time, chronic inflammation, formation of ulcers, and thickening of the intestinal wall occurs and leads to the signs and symptoms of the disease.
Crohn’s disease tends to run in families. Patients have a 5-10% chance that a first-degree relative, such as a parent or sibling, also has the condition. Compared to the other common IBD, ulcerative colitis, there is an increased risk of developing Crohn’s disease through genetic predisposition.
Overall, the risk of developing Crohn’s disease increases if both parents have IBD (7-9% if one parent has it vs 35% if both parents do).

The risk of developing either IBD is much higher when both parents have it. And it is more common among those with an eastern European background.
Diagnosis
The National Institute of Diabetes and Digestive and Kidney Diseases describes how to diagnose Crohn’s disease. The first step is normally a physical exam in which a doctor will check for signs of the disease in the abdomen. This will include checking for bloating, listening to the abdomen with a stethoscope, and checking for signs of an enlarged liver or spleen.
If Crohn’s disease is suspected, doctor’s will perform additional diagnostic tests.
Lab tests
Blood tests: Doctors draw a blood sample to check for red and white blood cells. Fewer than normal red blood cells is a common sign of anemia. On the other hand, a higher than average white blood cell count can be a sign of inflammation. This inflammation may or may not be related to Crohn’s disease.
Stool test: For this test, you provide a stool sample. Doctors analyze this for other potential causes of digestive issues, such as parasites.
Procedures
Internal endoscopies are the most accurate methods to diagnose Crohn’s disease and to rule out other conditions such as ulcerative colitis, diverticular disease, or cancer.
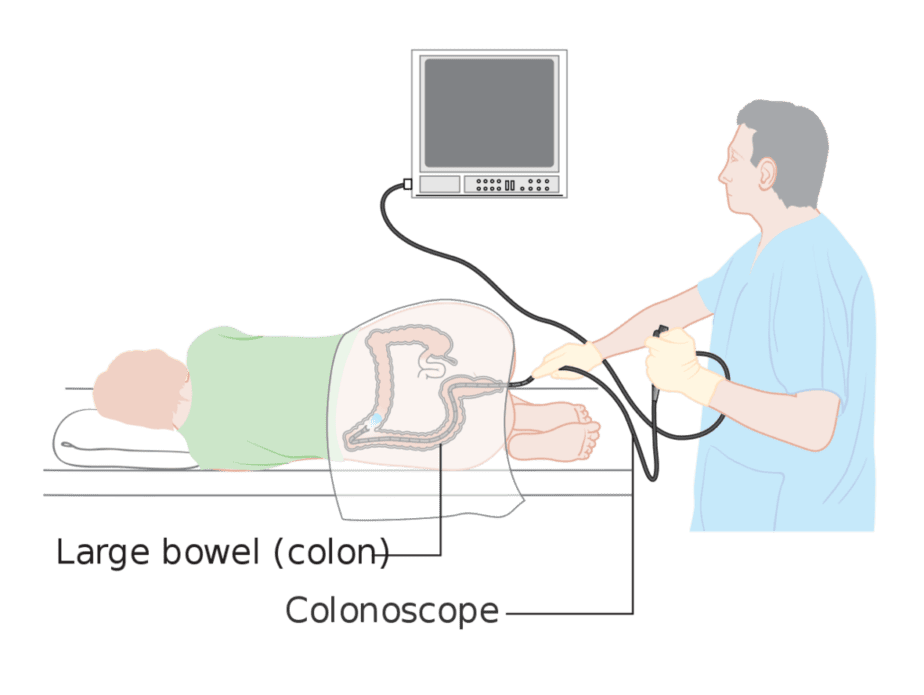
Colonoscopy: During this procedure, a specialist uses a long, flexible tube with a light and tiny camera on the end to observe your rectum and colon. Patients will follow bowel prep instructions and remain under anesthesia or pain medications during the procedure. You will lie on your left side on an exam table with your knees drawn to your chest while the specialist guides the camera through your anus and into the lower part of the ileum, the deepest part of the small bowel. They may also perform biopsies of the ileum, colon, and rectum at this time.
Upper GI endoscopy and enteroscopy: During an endoscopy, the doctor uses an endoscope to view the upper digestive tract. Unlike the colonoscopy, the technician inserts the camera down your esophagus and into your stomach and duodenum. They often administer liquid anesthetics for the throat and light sedatives with this procedure.
For enteroscopy, they use a similar procedure to observe part of the small intestine. In this case, the endoscope is longer to reach this area. Examples of this procedure include:
- push enteroscopy – a long endoscope examines the upper portion of your small intestine
- single- or double-balloon enteroscopy – small balloons to help move the endoscope into your small intestine
- spiral enteroscopy – a tube attached to an endoscope that acts as a corkscrew to move the endoscope into your small intestine
Capsule endoscopy: Patients swallow a capsule containing a tiny camera that the doctor uses to observe the GI tract. Unlike the other endoscopic procedures, patients do not need anesthetics.
Imaging
X-ray: Doctors can use x-rays to view the upper GI tract. During this procedure, the patient drinks barium while positioned behind the x-ray machine. Barium is a chalky liquid that makes the upper GI tract more visible to x-ray and fluoroscopy, which is often used at the same time.
CT scan: This scan takes images of part of the digestive tract. Patients will usually drink a special solution and receive an injection of contrast agent. They will then have pictures taken of the affected areas using an x-ray machine.
Treatment
There is no cure for Crohn’s disease, but there are treatment options to help manage symptoms. Treatment will be different for each patient and will largely depend on the types and severity of symptoms.
Medications
Anti-inflammatory drugs: These medicines are usually the first step to treating any inflammatory bowel disease. Your doctor may recommend drugs such as corticosteroids or oral 5-aminosalicylates. Doctors use the former strategically, prescribing it for only a few months to induce remission or in combination with an immune system suppressor.
Immune system suppressors: These target the immune system directly. A combination of these drugs often produce better results. Some examples of these medications are azathioprine, mercaptopurine, and methotrexate. The first two are the most commonly prescribed but may cause side effects such as nausea and vomiting or serious effects such as lowered resistance to infection and inflammation of the liver. Doctors typically prescribe methotrexate to those for which other medications did not work. Its side effects should be closely monitored.
Biologics: A relatively new type of medication, biologics target proteins made by the immune system. Natalizumab (Tysabri) and vedolizumab (Entyvio) stop integrins, a molecule on immune cells, from binding to other cells in the intestinal lining. Doctors prescribe the former less often due to risk of a serious brain disease that often leads to death or severe disability. The latter is a newer version of the medicine that does not have this risk.
Infliximab (Remicade), adalimumab (Humira), and certolizumab pegol (Cimzia) neutralize an immune system protein known as tumor necrosis factor (TNF). Finally, the FDA approved ustekinumab (Stelara) to interfere with interleukin, a protein associated with inflammation.
Antibiotics: These medications may reduce the amount of drainage from fistulas and abscesses and sometimes heal them. Due to their ability to reduce harmful bacteria, some doctors also believe they may reduce inflammation caused by infection.

Other commonly recommended medications include antidiarrheals, pain relievers, vitamins, and supplements.
Diet
Eating an intentional diet is an important component to treating Crohn’s disease through reducing symptoms and replacing lost nutrients. Many providers recommend keeping a food diary to note which types of food cause the most digestive distress.
Nutrition is especially important as many patients experience loss of appetite due to the digestive issues along with increased energy requirements due to presence of the disease. Common symptoms such as diarrhea further reduce the body’s ability to absorb necessary components from food such as protein, fat, and carbohydrates.
Sometimes, doctors recommend alternative nutritional intake administered through a feeding tube or injection to give the bowels a rest and reduce inflammation in the short term. They may prescribe this prior to surgery or when other methods fail to control symptoms.
Some other common diets include those that are low residue or low-fiber so as to reduce the risk of intestinal blockage (in the case of narrowed intestines, strictures), soft, bland diets, or reduction in dairy intake.
Surgery
If the condition does not respond to medications or if you develop a fistula, fissure, or intestinal obstruction, surgery may be recommended. Surgery does not cure the disease even though over half of patients will require at least one surgery in their lifetime. These procedures normally involve the surgeon removing the damaged areas of the digestive tract and reconnecting the healthy parts. Surgeries may also be used to close fistulas and drain abscesses. It often increases the quality of life for patients for years.
Unfortunately, symptoms of the condition often recur later in life near the area of reconnected tissue. When this happens, patients may undergo a second surgery. Following up the procedure immediately with medications can help increase the healing benefits.
Support from family members and friends is an important component to help patients deal with the mental distress caused by the disease.
If you liked this article, you should check out our other posts in the Nebula Research Library!
September 23, 2022
